HCC and the hepatic vascular niche: Analysis of angiocrine and adhesive vascular targets
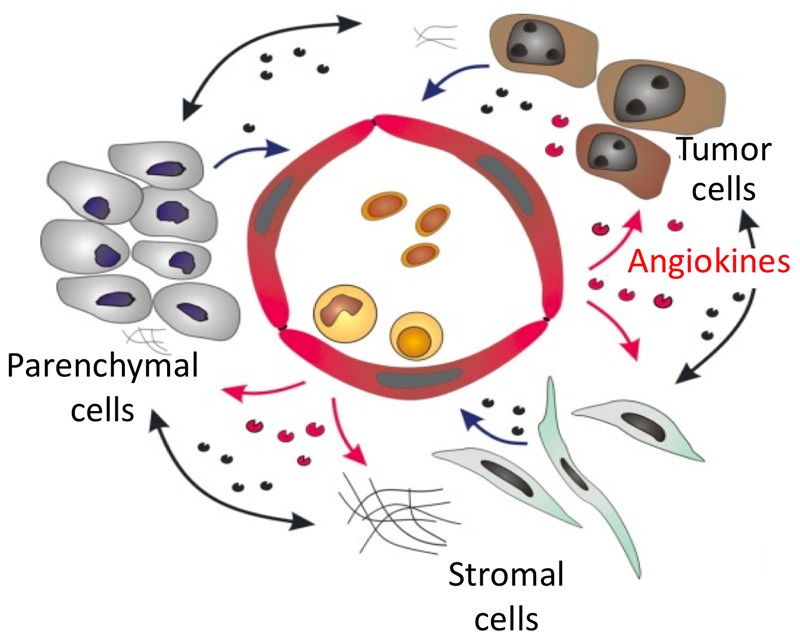
The angiogenic activity of hepatocellular carcinoma (HCC) has been correlated with high risk of microvascular invasion, metastasis and poor prognosis. Correspondingly, HCC is among the few tumor types in which anti-angiogenesis has some efficacy as monotherapy. Yet, the overall clinical efficacy is limited and the molecular mechanisms of HCC angiogenesis remain poorly understood. Moreover, no treatment is capable to reduce the high frequency of post-resection HCC recurrence that remains a major clinical concern for the prognosis of HCC. The intrahepatic sinusoidal vasculature has unique organ-specific characteristics with a dual arterial/venous blood supply. It is therefore conceivable that tumor angiogenic processes in HCC are mechanistically different from angiogenesis in other solid tumors including hitherto poorly understood processes of vascular reprogramming termed “capillarization”. To systematically study the mechanisms and therapeutic potential of HCC angiogenesis, the two partnering laboratories have joined forces to exploit their labs’ specific expertise in liver sinusoidal endothelial cell (LSEC) biology (Goerdt laboratory) and tumor angiogenesis (Augustin laboratory) towards a concerted effort to study tumor-vessel interactions during HCC progression and postsurgical recurrence. On the basis of this collaboration, we have thoroughly studied (i) the molecular repertoire of LSEC including angiocrine regulation of liver homeostasis and hepatocyte proliferation as well as (ii) hepatic microvascular reprogramming during hepatocarcinogenesis in a novel preclinical HCC model suitable for proof-of-concept therapy experiments (iAST model). These findings straightforwardly guide the experimental program of project C3: First, we propose to mechanistically study the functional crosstalk between LSEC and HCC cells concentrating on LSEC-specific angiokines (Wnt, Bmp2 and HGF) as well as on candidate molecules for liver-specific HCC-EC adhesion and transmigration (stabilin 2, LYVE-1, and CD44).Second, we propose to identify and validate known and novel angiocrine and angiogenic factors as vascular targets in HCC and to exploit the therapy-validated iAST model for the systematic and rational preclinical development of novel angio-directed combination therapies. The iAST model will towards this end be advanced to a locally induced tumor model that upon primary tumor removal allows the study of metastasis and recurrence providing the first mouse model recapitulating post-surgical recurrence of human HCC. This advanced tumor model coupled to above mentioned pre-clinical studies (combination therapies) will enable the assessment of the functional role of different candidate molecules in promoting HCC recurrence. Together, the experimental program aims to unravel the complexity of tumor-vessel crosstalk during HCC angiogenesis and microvascular invasion for the preclinical validation and proof-of-principle exploitation of novel therapeutic targets.